The Clinical Trials Directive: How Is It Affecting Europe's Noncommercial Research | PLOS Clinical Trials
E17 General Principles for Planning and Design of Multiregional Clinical Trials Guidance for Industry
Large Clinical Trial of Hydroxychloroquine for COVID-19 Sponsored by Novartis After Reaching Agreement With FDA

Comparison of clinical trial guidelines in USA, EU and India, Singapore. | Download Scientific Diagram
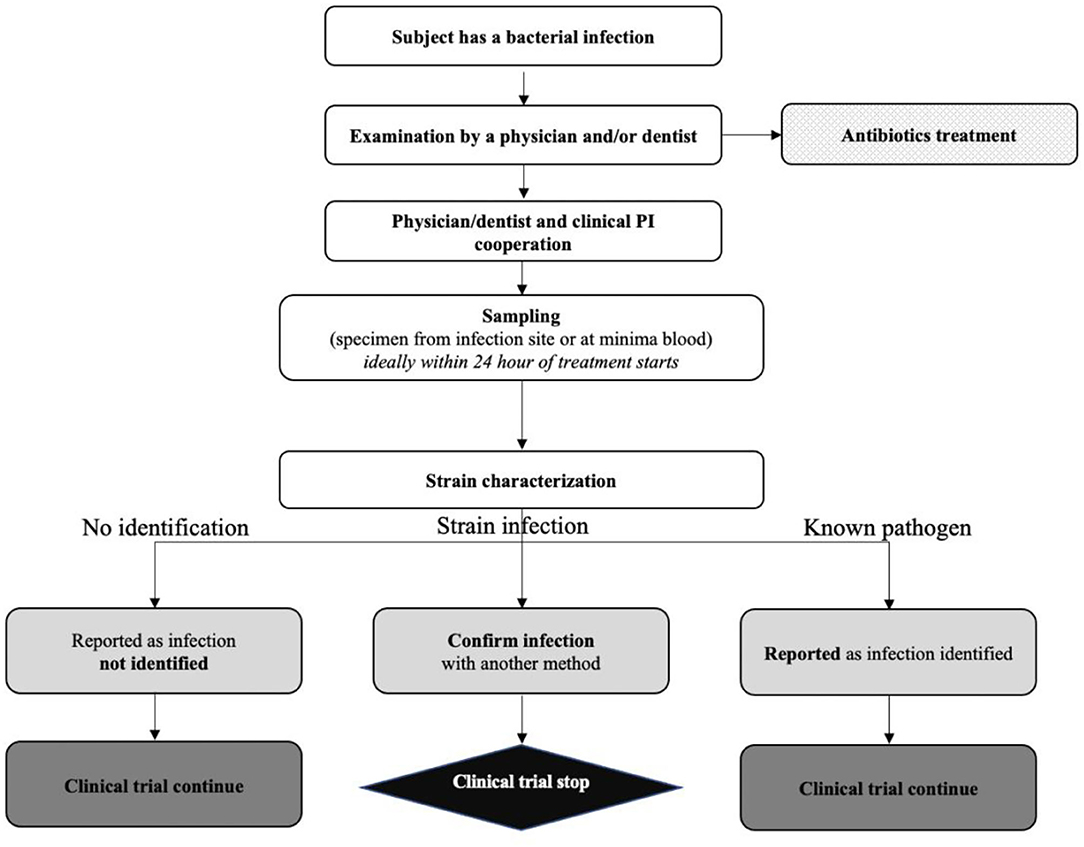
Frontiers | Entering First-in-Human Clinical Study With a Single-Strain Live Biotherapeutic Product: Input and Feedback Gained From the EMA and the FDA

Incorporating Site-less Clinical Trials Into Drug Development: A Framework for Action - Clinical Therapeutics