
Clinical Trials Management Certificate — online course - University of Delaware Div. of Professional and Continuing Studies

Clinical Research Equity Agency - Clinical Data Managers are earning an average of $78,174/year! You can become one in 2 months. Study online, take your certification exam in your hometown and become

Principal Clinical Data Manager - Multiple Sponsors, Late Phase job with Labcorp Drug Development | 1438026

Data Management & Case Report Form Development in Clinical Trials. Introduction to the Principles and Practice of Clinical Research. - PDF Free Download

Principal Clinical Data Manager - Medical Devices & Diagnostics job with Labcorp Drug Development | 1422841
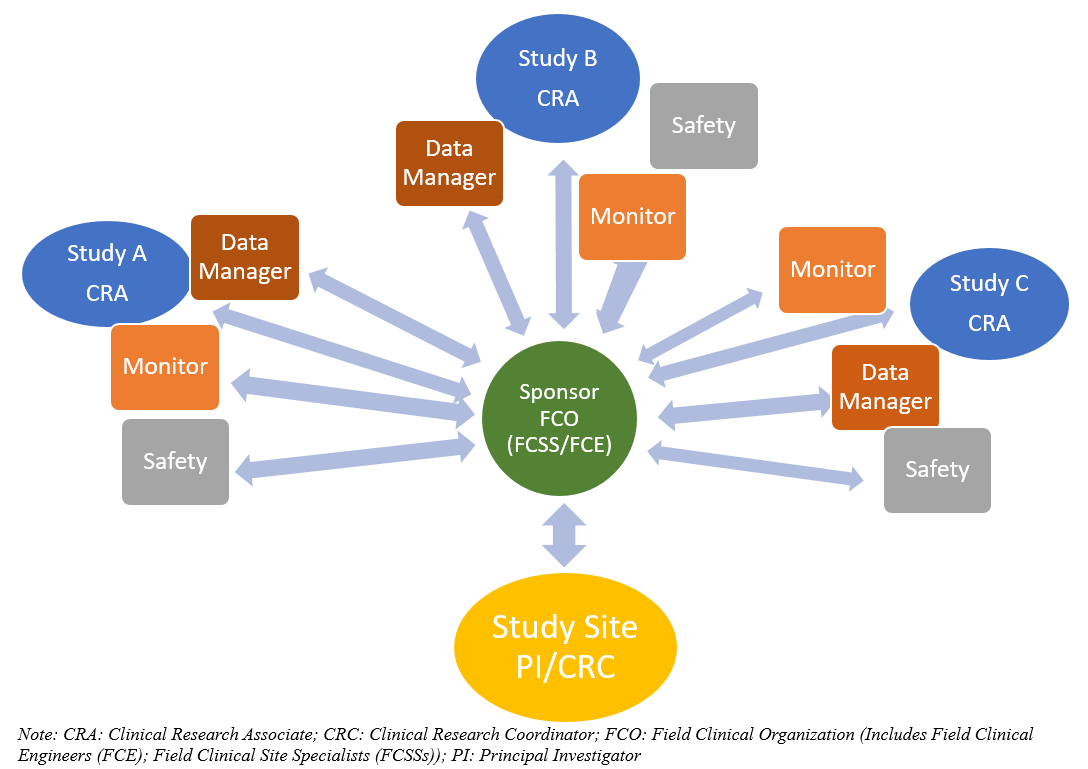
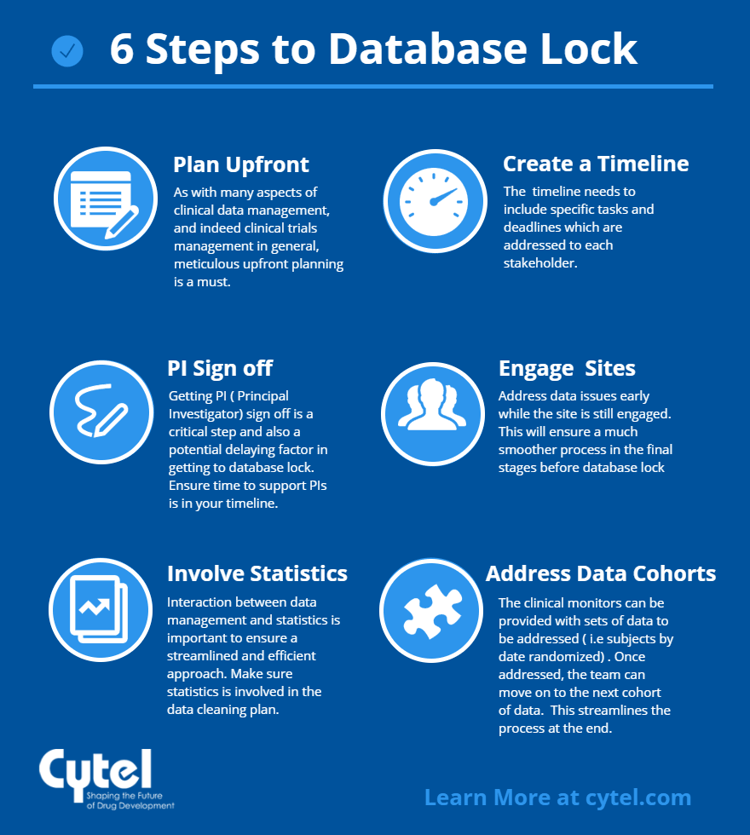




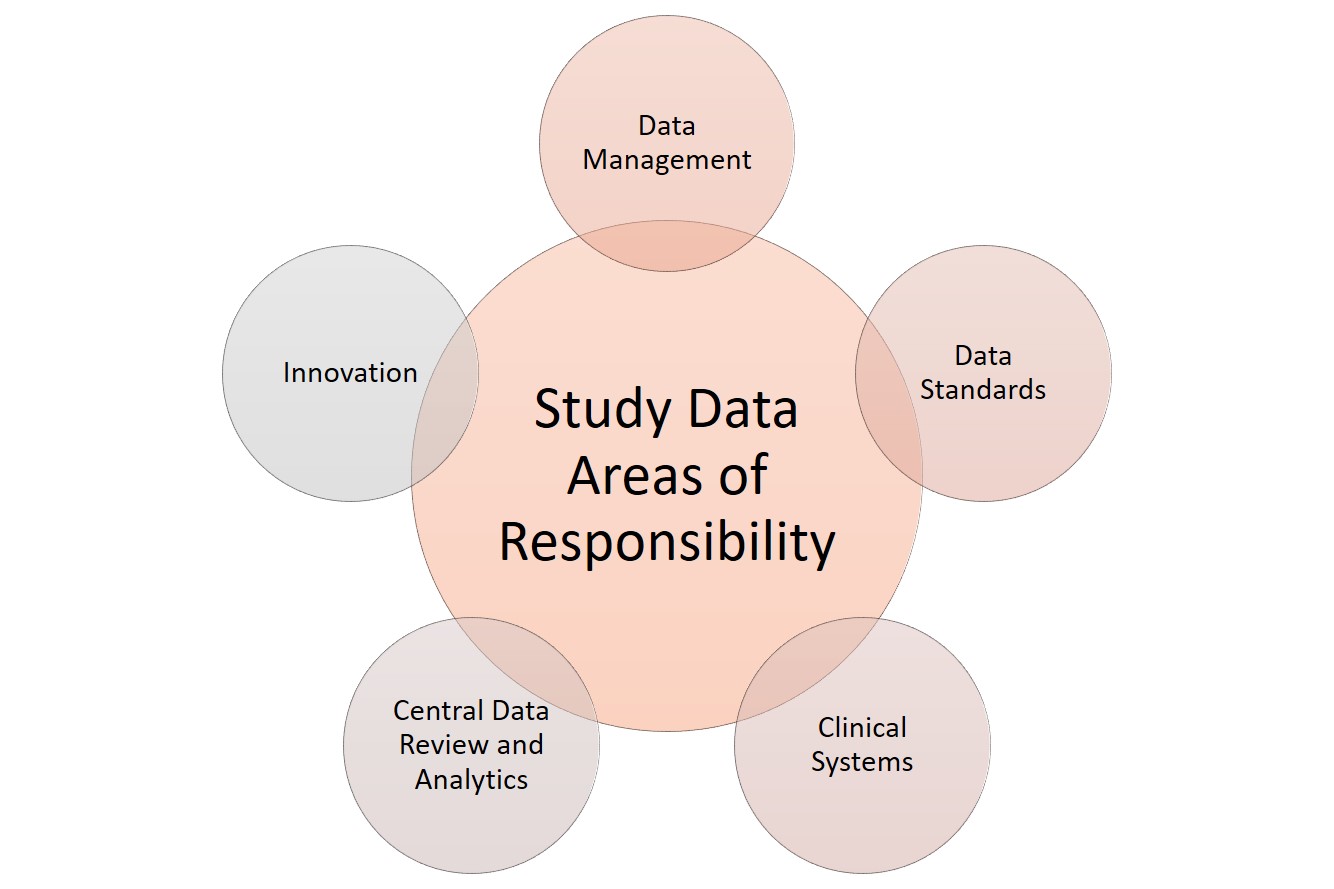


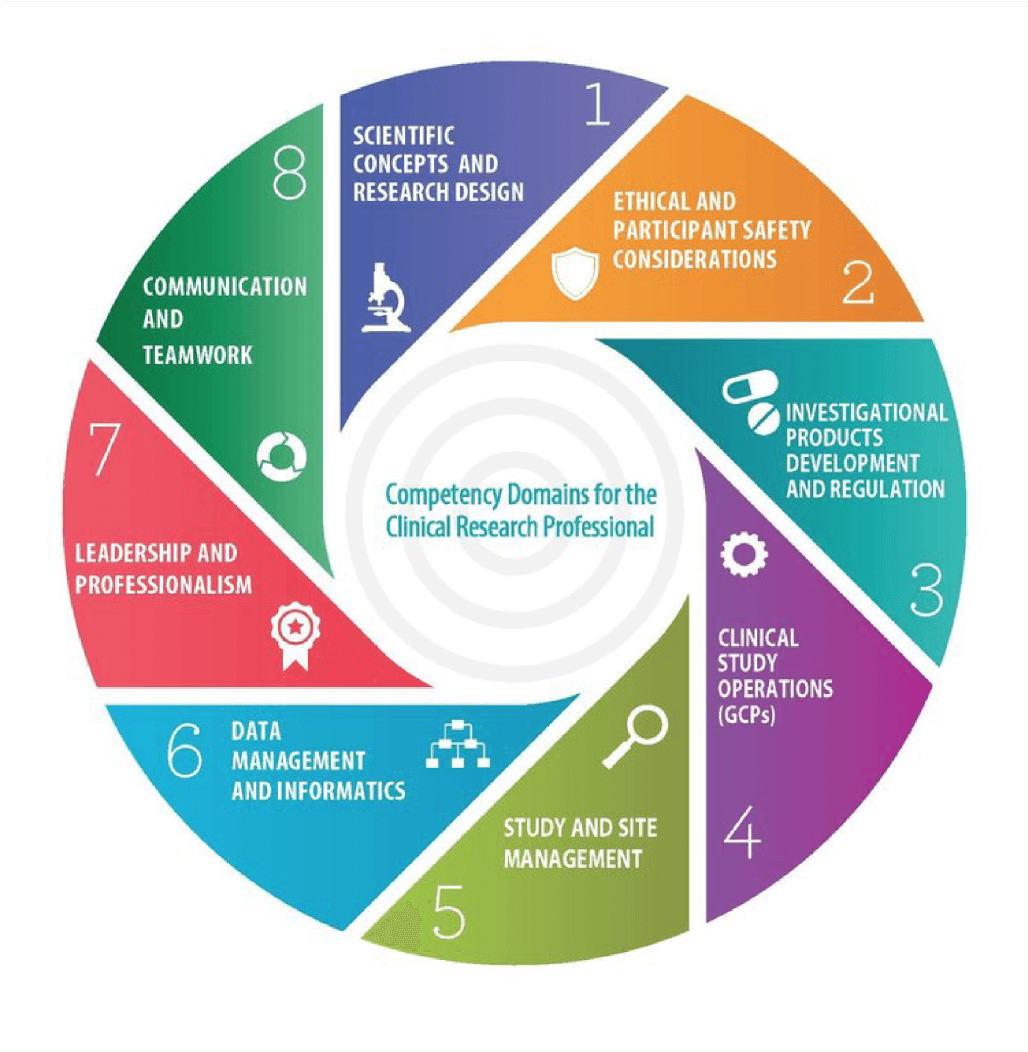
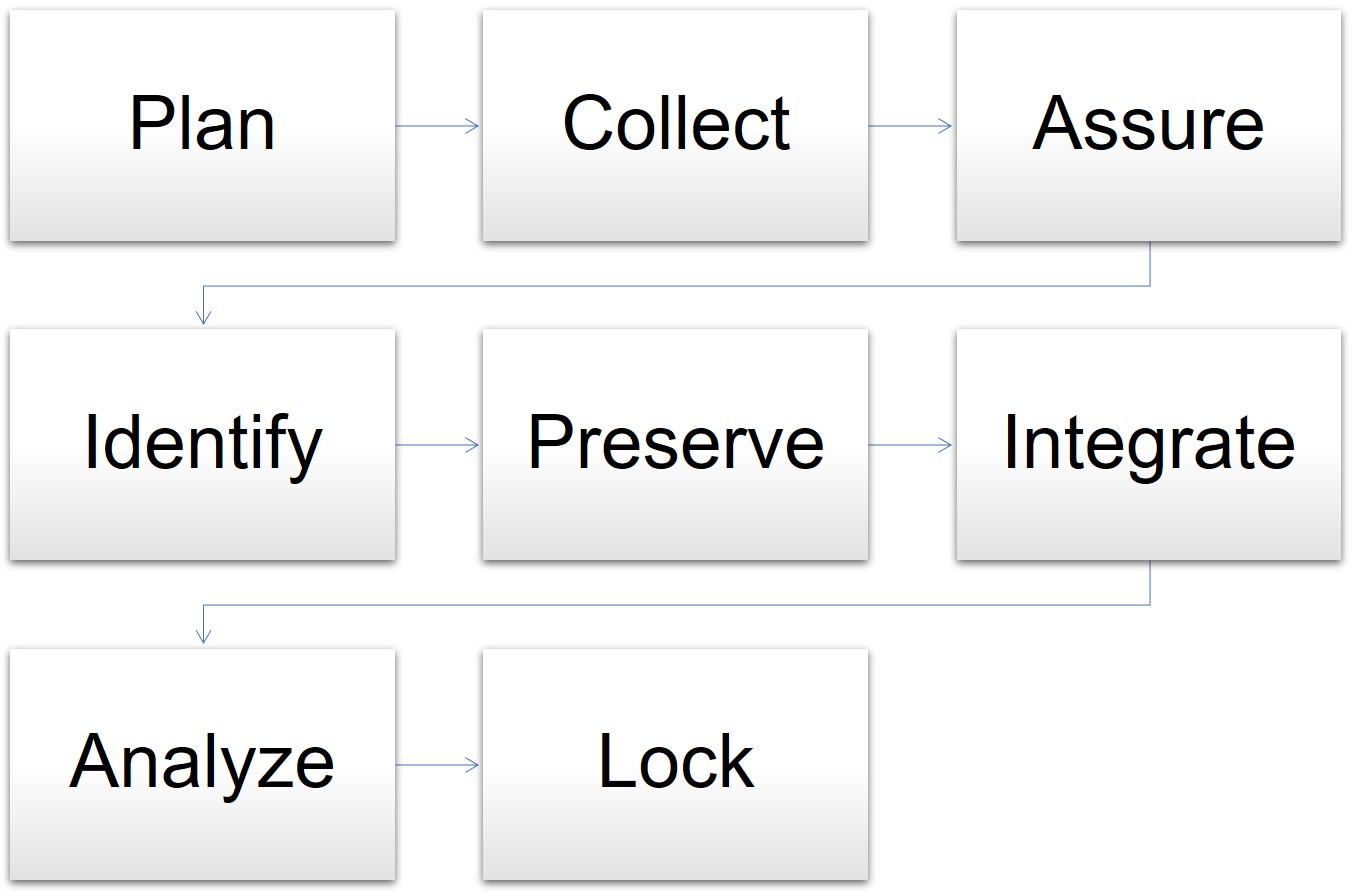
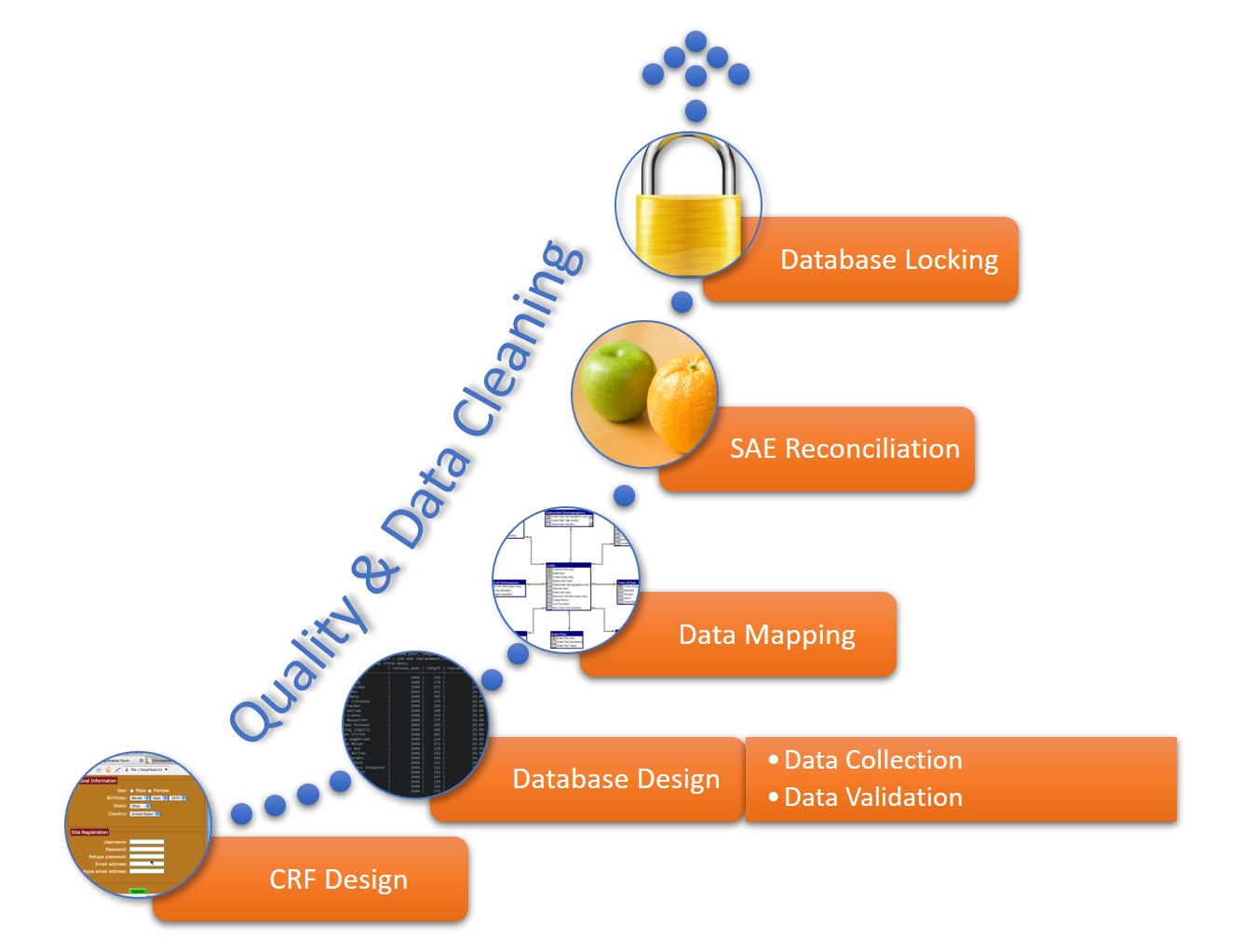

.png)



